Thomas Beneyton
Thomas Beneyton- PhD Student started October 2008.
Project
Digital microfluidic systems for the directed evolution of catalysts for biofuel cells
Our final goal is to build a functional bio-fuel cell using enzymes as redox catalysts at the anode and cathode. To achieve this goal the key points are a high catalytic activity of these enzymes at high ethanol concentrations, broad pH and temperature variations and their stability at the interface with the electrodes. These are conditions under which the natural enzyme will never evolve in vivo. Hence, we are aiming to apply in vitro protein evolution methods in combination with digital microfluidics to create better catalysts for the "green energy" production.
The expenses concerning "green energy" production might be overcome by availability of better bio-catalysts. In the most common way biofuel, in form of ethanol, is obtained from sugars stored within the plants such as corns, sugarcane.
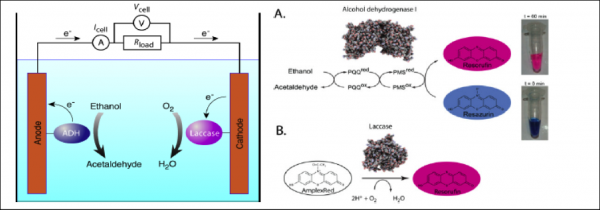
Schematic configuration of a biofuel cell based on redox enzymes and the fluorogenic tests for measuring the activity of the biocatalysts in droplet: (On the left) ethanol is oxidized to acetaldehyde by alcohol dehydrogenase (blue ellipse). During oxidation, electrons from ethanol are transferred onto the anode via PQQ cofactor, while protons are released into the media. At the cathode, electrons are transferred to immobilized laccase (purple ellipse), which catalyzes oxygen reduction to water in the presence of copper ions. (On the right) panel A: the fluorogenic assay for the PQQ-ADH: in the presence of ethanol, the Pyrroloquinoline quinine (PQQ) cofactor and the PMS (Phenazine MethoSulfate: electron mediator), resazurin is converted to resorufin. (On the right) p anel B: the fluorogenic assay for the laccase: in the presence of oxygen, AmplexRed is converted to resorufin. Taken from Skhiri, Y., Beneyton, T. et al., Proceedings MicroTas 2010.
For a successful evolution laccase using digital microfluidics, my project consisted on addressing the following challenges:
- Development of new tools and procedures for the immobilization and electrochemical analysis of CotA, a bacterial extremophilic enzyme that has not previously be used for biofuel cell applications
- Creation of reliable microfluidic platforms for directed evolution of laccase
- Optimisation of Cot A for biofuel cell applications
Publications
Immobilisation of CotA, an extremophilic laccase from Bacillus subtilis, on glassy carbon electrodes for biofuel cell applications. Beneyton, T., El Harrak, A., Griffiths, A.D. and Taly, V. Electrochemistry Communications (2011), 13(1): 24-27.
The thermophilic CotA laccase from Bacillus subtilis: Bioelectrocatalytic evaluation of O2 reduction in the direct and mediated electron transfer regime. Beneyton, T., Beyl, Y., Guschin, D., Griffiths, A.D., Taly, V. and Schuhmann, W. Electroanalysis (2011), 22: DOI: 10.1002/elan.201100054.
Hybrid Bidentate phosphoramidite ligands in asymmetric catalysis. Boeda F., Beneyton T. and Crevisy C. Mini-reviews in organic chemistry (2008), 5(2): 96-127, DOI: 10.2174/157019308784223613.
Proceedings
Integrated microfluidic platform for directed evolution of laccase for Biofuel cell applications. Skhiri, Y., Beneyton, T., El Harrak, A., Griffiths, A.D. and Taly, V. In: Proceedings of the 14th International Conference on Miniaturized Systems for Chemistry and Life Sciences (MicroTAS), 2010, p. 980-982. ISBN: 978-0-9798064-3-8.
Curriculum Vitae
Fundings
Région Alsace/ ADEME (Agence de l'Environnement et de la Maîtrise de l'Energie, http://www2.ademe.fr/servlet/getDoc?id=11433&m=3&cid=96)
Collaborations
Prof. P. Hellwig (see http://www-chimie.u-strasbg.fr/~lsveb/ )
Prof. Dr. Wolfgang Schuhmann (http://www.ruhr-uni-bochum.de/elan/ )
Dr J.-C. Baret (http://dmi.ds.mpg.de/ )